Drs. Joyce and Jani look toward the future of liquid biopsy in RCC care and how new technology, like AI, will play a role.
Renal Cell Carcinoma Diagnostics
Advertisement
Drs. Joyce and Jani discuss practical clinical cases of using liquid biopsy to manage patients in different RCC settings.
Drs. Joyce and Jani talk about the technique of liquid biopsy and its role as a diagnostic tool in RCC.
Dr. Machaalani highlights his study that evaluated the association between circulating KIM-1 and mRCC tumor biology.
High KIM-1 may be a biomarker of poor prognosis in RCC and correlates with specific LOF mutations, transcriptions programs.
68Ga-NY104 detected using PET/CT shows better diagnostic efficacy than 18F-FDG PET/CT in patients with mccRCC.
Drs. Ben-David and Joyce evaluate the performance of ctDNA in detecting localized RCC disease recurrence after surgery.
Drs. Ben-David, Joyce share an analysis on recurrence-free survival according to ctDNA status in patients with renal masses.
Pre-op ctDNA in localized RCC is linked to aggressive tumor features and worse RFS, highlighting its prognostic potential.
CA9 imaging and ZIRCON data offer new insight into managing small renal masses and supporting personalized kidney cancer care
Drs. Joyce and Chang discuss diagnostic strategies and future innovations in managing small renal masses.
Video Insights
Conferene Coverage
Samer Srour, MB, talks about the updated ALLO-316 and its use in targeting CD70 in patients with advanced ccRCC.
Dr. Flavell discusses how landmark studies have shaped current trial designs, and growing interest in combination therapies.
Dr. Flavell highlights the expansion of lutetium PSMA therapies into earlier disease settings: HSPC and oligometastatic PC.
Preliminary trial data suggests that 18F-DCFPyL PSMA PET/CT combined with mpMRI may improve csPCA detection over MRI alone.
Quantitative analysis outperformed visual analysis in the prediction of outcomes for patients undergoing Lu-PSMA therapy.
New research explores the prognostic potential of LuPSMA-SPECT/CT with RECIP 1.0 in patients with advanced prostate cancer.











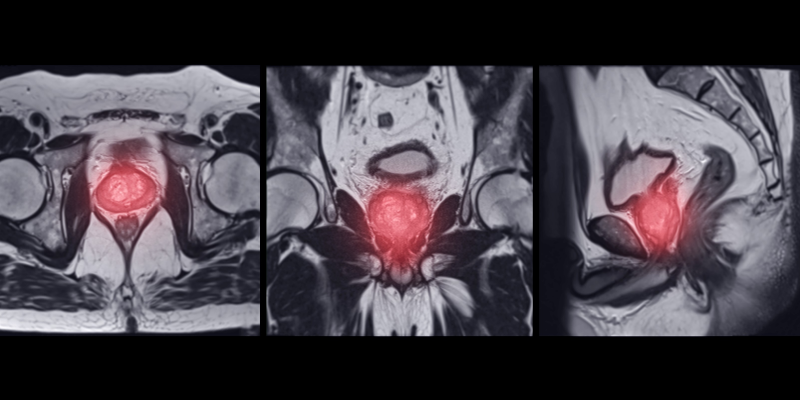
![Monitoring [177Lu]Lu-PSMA Therapy in mCRPC: Clinical Protocols](https://mumcdnstorage.blob.core.windows.net/guonc/2022/09/Metastatic-prostate-cancer.jpg)


 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.