
mCSPC
Advertisement
Drs. Swami and Ciuro discuss efforts to reduce disparities in the real-world application of mHSPC intensified therapy.
Dr. Attard explores how the study's results compare with prior data in the metastatic castration-resistant setting.
Adding niraparib to abiraterone and prednisone prolongs rPFS in patients with mCSPC harboring HRR gene alterations.
Drs. Ong and Roy detail a multinational real-world analysis of the IRONMAN registry on the significance of PSA>0.2.
Drs. Swami and Ciuro discuss adoption of treatment intensification, barriers to guideline-concordant care, and more in mHSPC.
The approval of darolutamide by the FDA expands treatment options available for patients diagnosed with mCSPC.
Drs. Privé and Tagawa discuss the BULLSEYE trial, an analysis of Lutetium-177-PSMA-617 in oligometastatic HSPC.
Researchers evaluated the addition of zoledronic acid to the standard treatment of enzalutamide and ADT.
Drs. Swami and Ciuro discuss real-world data that indicates a shift in mHSPC treated with intensified therapy since 2023.
Dr. Shore highlights post hoc analyses of ultra-low PSA responses and their correlation with outcomes in the ARANOTE study.
A recent study evaluated the impact of early withdrawal of upfront ARSIs on clinical outcomes in patients with mCSPC.
Patients with ARPI-treated mCSPC and BRCA1/2 alterations experienced higher rates of disease progression.
Dr. Antonarakis discusses the differences between PARP1 mRNA expression and other biomarkers used for risk stratification.
Dr. Ciuro discusses key factors to consider when choosing between doublet and triplet therapy for mHSPC.
The panel highlights the limitations of conventional imaging in detecting disease progression at undetectable PSA levels.
The panel zooms in on the use of radium, enzalutamide, and other therapeutic agents, touching on patient preference trials.
The panel focuses on decision-making between AR antagonists and abiraterone, weighing factors such as reimbursement.
The panel goes in-depth on the use of testing and SBRT as metastasis-directed therapy in newly diagnosed prostate cancer.
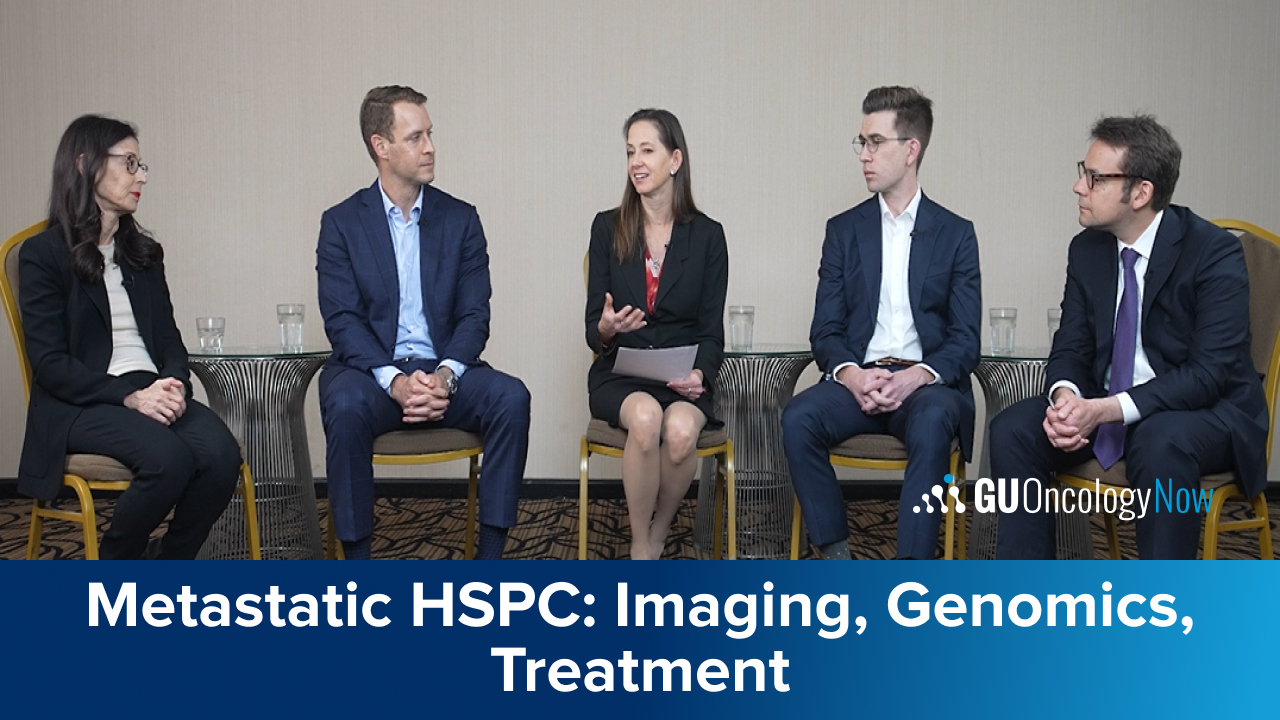
The panel explores the the role of imaging, genomics, and patient factors in the management of mHSPC.
In 53.3% of cases, PSMA PET positively identified bone disease despite negative findings on bone scintigraphy.

















 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.