
ESMO 2022
European Society for Medical Oncology Congress 2022
Advertisement
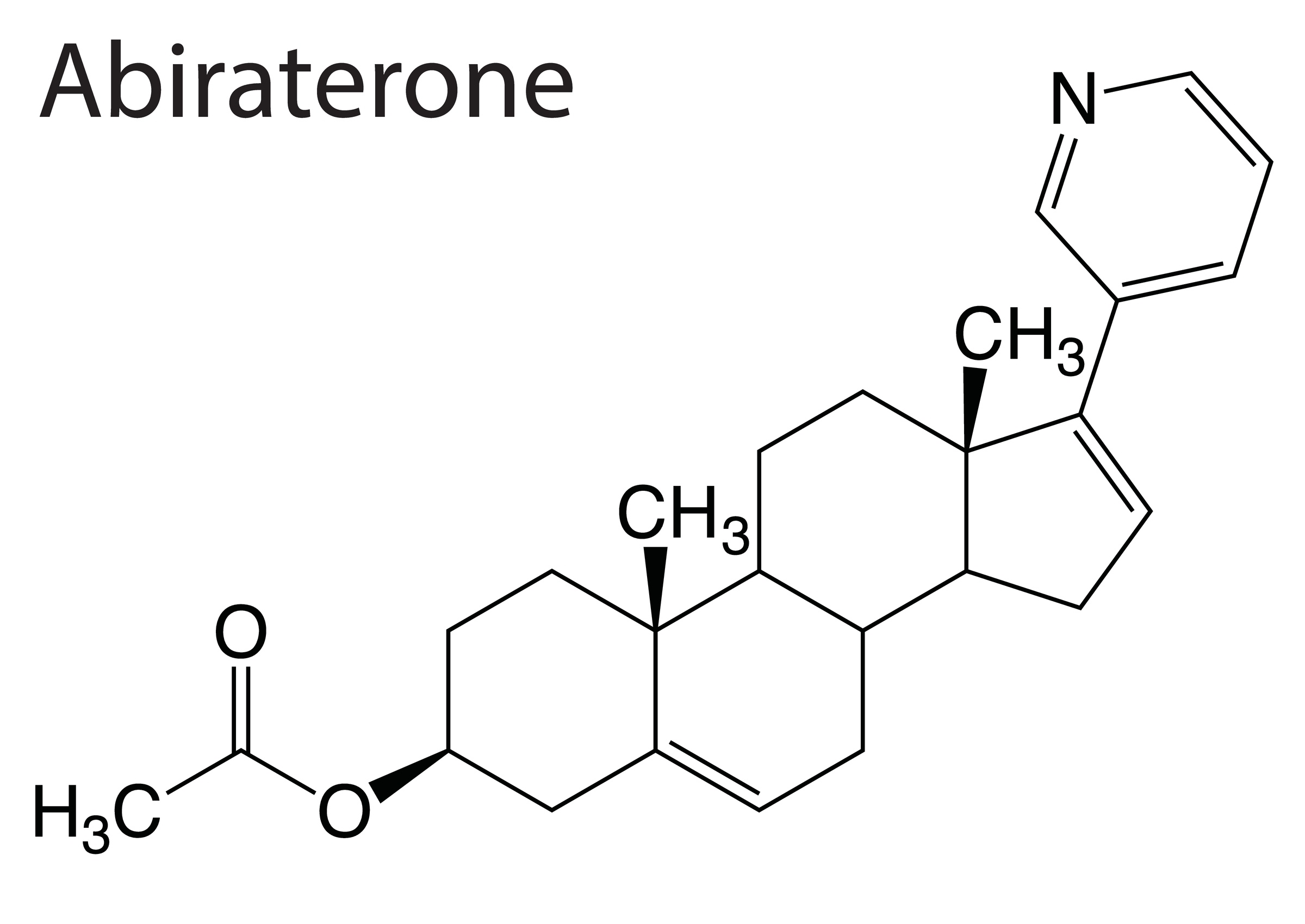
Abiraterone acetate + prednisolone is compared to enzalutamide + AAP for mHSPC cancer starting androgen-deprivation therapy.
Results from the phase 3 PROpel trial on abiraterone + olaparib vs abiraterone + placebo as first-line therapy for MCRPC
Neoadjuvant platinum-based chemotherapy has been found effective for treating metastatic penile squamous cell carcinoma.
A new study has found an association between neurodevelopmental disorders and testicular seminoma.
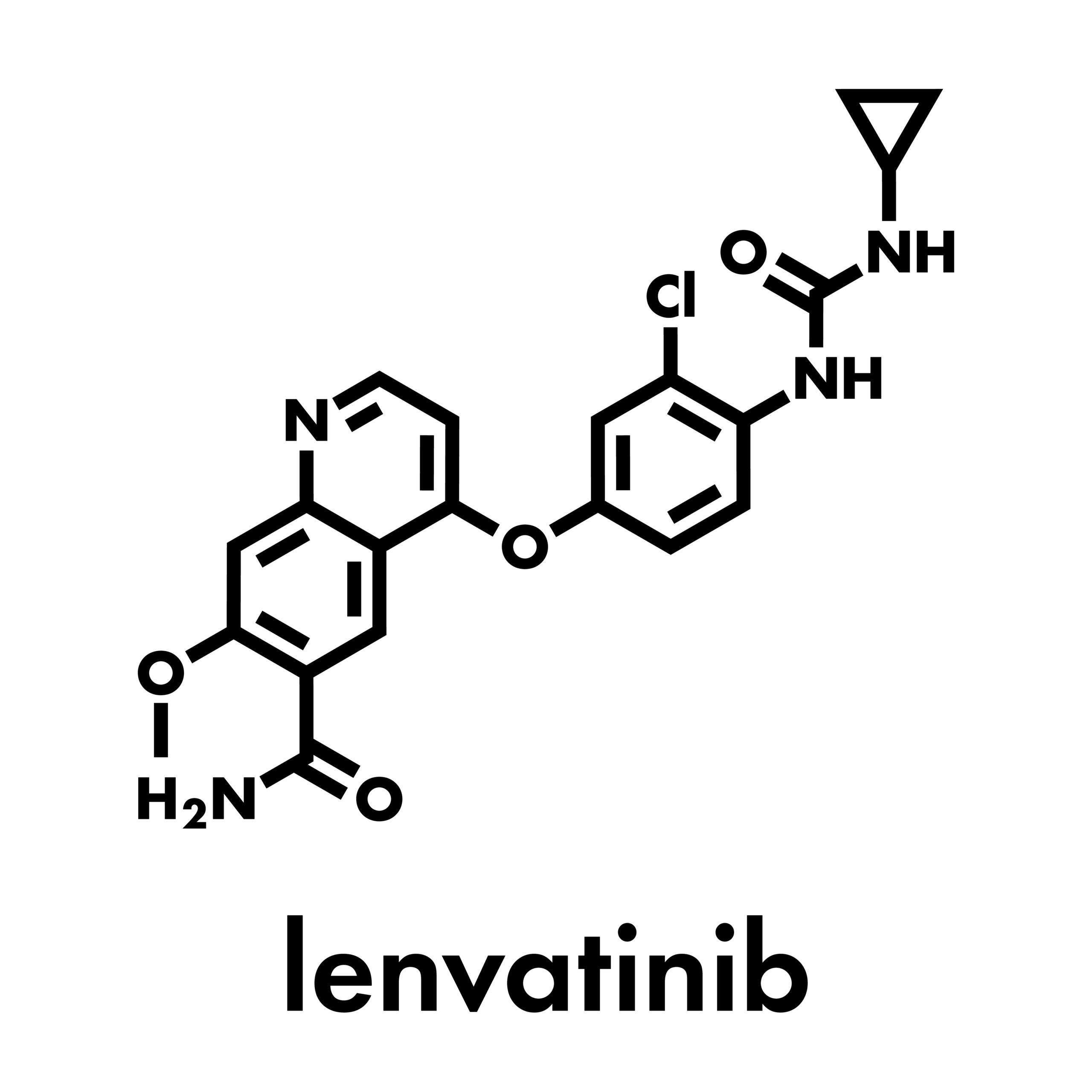
CLEAR study results showed that treatment with first-line lenvatinib plus pembrolizumab had clinical benefit at 2 years.
The RACE IT trial finds neoadjuvant radio-immunotherapy followed by radical cystectomy a safe and feasible treatment option.
Patients with mCRPC with AR alterations had significantly worse survival outcomes compared with patients with wild-type.
A meta-analysis found that triplet regimens with darolutamide and APP improved survival in mCSPC with high volume of disease.
Pembro plus AAP sustained antitumor activity with acceptable safety signals in patients with mCRPC after 2 years.
PRESTO trial results show adding APA and AAP to ADT improved biochemical PFS in patients with relapsed prostate cancer.
ESMO 2022: Enfortumab Vedotin Plus Pembrolizumab Versus Pembrolizumab Alone for Urothelial Carcinoma
Peter O'Donnell describes the randomized phase 2 study on enfortumab vedotin and pembrolizumab against pembrolizumab alone.177Lu-PSMA-RLT showed efficacy and safety consistent with published evidence in cohort of very old patients with mCRPC.
Karim Fizazi discussed preliminary findings on ODM-208, a first-in-class CYP11A1 inhibitor at ESMO 2022.
Late-breaking data from 2 randomized phase 3 trials that followed STAMPEDE protocol were presented at the ESMO Congress 2022.
Nizar Tannir describes this randomized phase 3 study on bempegaldesleukin plus nivolumab as presented at ESMO.
The phase III PRESTO study examined the effects of androgen suppressants on biochemically relapsed prostate cancer.
Axel Bex summarizes the ipi/nivo, pembro and atezo studies that were presented at ESMO 2022.
Mohamad Allaf gives a recap of the open-label PROSPER study on neoadjuvant nivolumab in RCC.
Researchers evaluated immune effects of enzalutamide in patients with metastatic castration-resistant prostate cancer on ADT.
Pembrolizumab plus olaparib did not improve survival versus alternating from abiraterone or enzalutamide in mCRPC.




 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.